The effect of Water Composition on the Absorption of C02 |
The Dissolving of Shells Due to a High Rate of CO2 |
|
Introduction: Ocean acidification is when CO2 from the atmosphere gets into the ocean and creates carbonic acid. As this continues, the ocean's acidity will double by the end of the century and will have a major effect of sea life (high acidity dissolves the shells of phytoplankton (key stone creatures).
Experimental Question: What happens when carbon dioxide is added to
seawater? Pre-Lab Questions:
1. What gad are you blowing into the water? We are blowing carbon dioxide into the water. 2. What happens to the gas when you blow it into the water? When you blow the gas into the water, the water absorbs it. 3. How are you measuring change in the water during this lab? We are measuring the change through the color. 4. What does the measuring of the pH of the water tell us? It tells us how acidic the water has become. 5. After studying the reactions above, how do you think carbonic acid will affect the pH of salt water? I think that carbonic acid will decrease the pH levels in salt water. Hypothesis: If I blow CO2 into the saltwater, then the saltwater's pH will decrease.
Protocol:
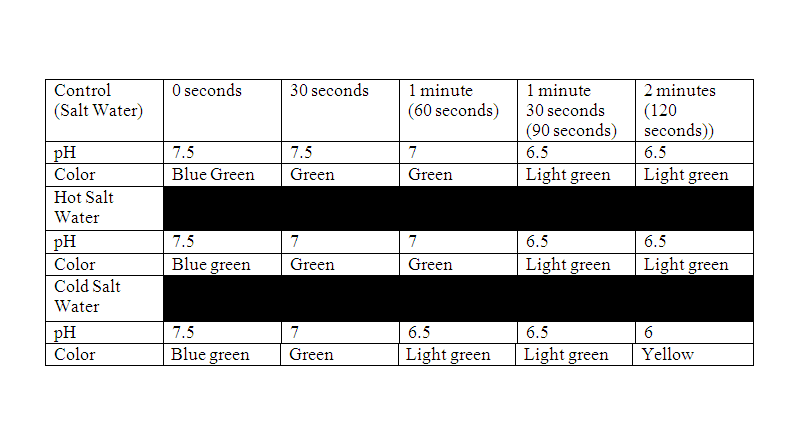
For the control of the experiment which was salt water we put 100 mL of salt water into a beaker. Then put 4 drops of the universal indicator into the salt water. After that we covered it with saran wrap and put a straw through it and blew into it for 2 minutes. For the second measurement we replaced the salt water with hot salt water. We also put the hot salt water into a beaker and added 4 drops of the universal indicator and covered it with saran rap. Then put a straw into it and blew on it for 2 minutes. Dependent Variable: The pH after the 2 minutes Independent Variable: The type of water Constant: The amount of water |
Introduction: Some organisms in the ocean have shells made of calcium carbonate (CaCo3). As the ocean becomes more acidic, those shells will dissolve because the organism is now absorbing more carbon the calcium carbonate and will have less energy to rebuild their shells. Since they will no longer have shells, those organisms will become exposed to predators.
Experimental Question: In which liquid will the shells dissolve in more quickly?
Pre-Lab Questions:
1. How do organisms make their shells? What are shells made of? They are made by putting calcium ions and carbonate ions from seawater making calcium carbonate. 2. What do you expect to happen to the shell in an acidic solution such as vinegar? I expect the shells to begin to dissolve. 3. What are sources of carbon dioxide and which of these sources are most likely to affect ocean pH? The burning of fossil fuels such as coal or oil Hypothesis: If a shell is put into vinegar, then the shell will dissolve.
Protocol:
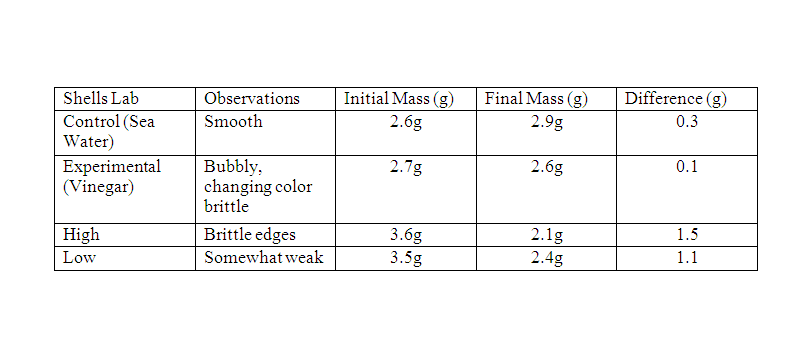
We started with finding the mass of the shells. The poured 150 ml of vinegar into a 500 ml beaker and 150 ml of salt water into another beaker. Then place the control shell in the salt water and add the experimental shell into the beaker of vinegar and leave it for 30 minutes. Then record the observations of the shells characteristics. After 30 minutes find the mass of both the control and experimental shell. Then test the strength of the shell. Dependent Variable: Mass of shells Independent Variable: Type of liquid Constant: The amount of liquid |
|
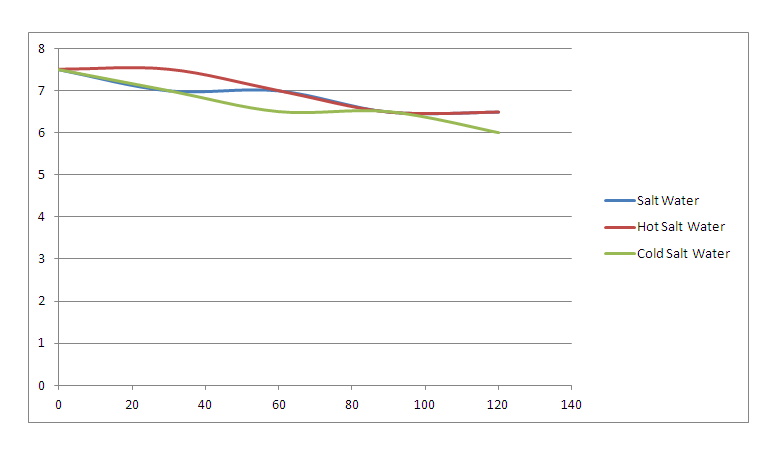
Data Analysis: 1. As you blew through the straw, what were you adding to the water and how did that change the pH?
The pH of the saltwater decreased. 2. What did the universal indicator tell us about the water? The universal indicator told the pH of the water. 3. What does this tell us about the effects of carbonic acid in the ocean water? When carbon goes into the ocean the sea water gets more acidic 4. Based on the results of your experimental protocol, which factor affects the pH of the water most, temperature or salt? Based on the results of experimental protocol, temperature affects pH of water the most. Conclusion: My hypothesis was correct, I if we blew carbon into the sea water that the pH would change. On the pH scale, any number below 7 is acidic. The pH of the salt water (control) started out as 7.5 and ended at 6. As I blew CO2 into the water, it got more acidic. This experiment was the same thing that's happening into the ocean. CO2 goes into the ocean from the atmosphere, it becomes carbonic acid making life for sea life harder. The sea life need a little carbon but not to much.
|
Data Analysis: 1. When you immersed the shells in vinegar how did you know that a reaction was happening?
I knew a reaction was happening when I seen the bubbles forming around it 2. How did observing the shells in vinegar relate to how animals are affected by a lower pH of ocean water? Watching the shells in vinegar shows how the sea animals are living. it shows that the acidic water was very harmful to their shells 3. How would shelled organisms be affected by a lower pH of ocean water? Shelled organisms will be affected by a lower pH of ocean water because their shell will dissolve. The lower the pH the more acidic the water is. 4. What are the primary functions of shell for these animals? The primary functions of a shell for these animals are for protection and shelter. 5. Does it cost the animal energy to rebuild or repair their shell? It does cost the animal energy to rebuild shells. Conclusion: This experiment showed that, on a global level, as more CO2 goes into the ocean and forms carbonic acid, the more the shells of some sea organisms will dissolve. As their shells dissolve, those organisms will lose their protection and shelter and be more exposed to predators. Without certain organisms in the ocean, it would alter the food chain in the ocean and how some of us humans live.
|